Stem cell procedures,
practiced under US law,
in five US states.
Select Stem Cell is a patient-services company. We connect adults with state-licensed physicians whose stem cell practices operate within the statutory frameworks of Florida, Texas, Utah, Wyoming, and Tennessee. The procedures discussed here are investigational and are not approved by the US Food and Drug Administration. Outcomes vary by patient and are not guaranteed.
05
States with Statutory Authority
12+
Indications Discussed
24h
Intake Review Window
Operating reference frameworks
For years, US patients had no statutory framework.
Now five states do.
In 2025, Florida enacted the Stem Cell Therapy Act (SB 1768). In 2026, Wyoming enacted the Stem Cell Freedom Act and Tennessee enacted HB 2246. Combined with Texas's investigational stem cell statute (HB 810/3148, often called "Charlie's Law") and Utah's amended consent framework (SB 199), five US states now have statutes that explicitly address how licensed physicians may perform investigational stem cell procedures.
Select Stem Cell is not a clinic. We are a patient-services company that helps adults understand the law in each state, prepare documentation, and connect with a licensed physician for a consultation. Any medical decision is made between the patient and the treating physician.
Read the state-by-state legal summaryFive states.
Each with explicit statutes.
We refer adults only to physicians practicing in states that have passed explicit legislation addressing investigational stem cell procedures. Each physician in our directory operates within their state framework, sources cells from FDA-registered, accredited establishments, and follows their state's informed-consent and disclosure requirements.
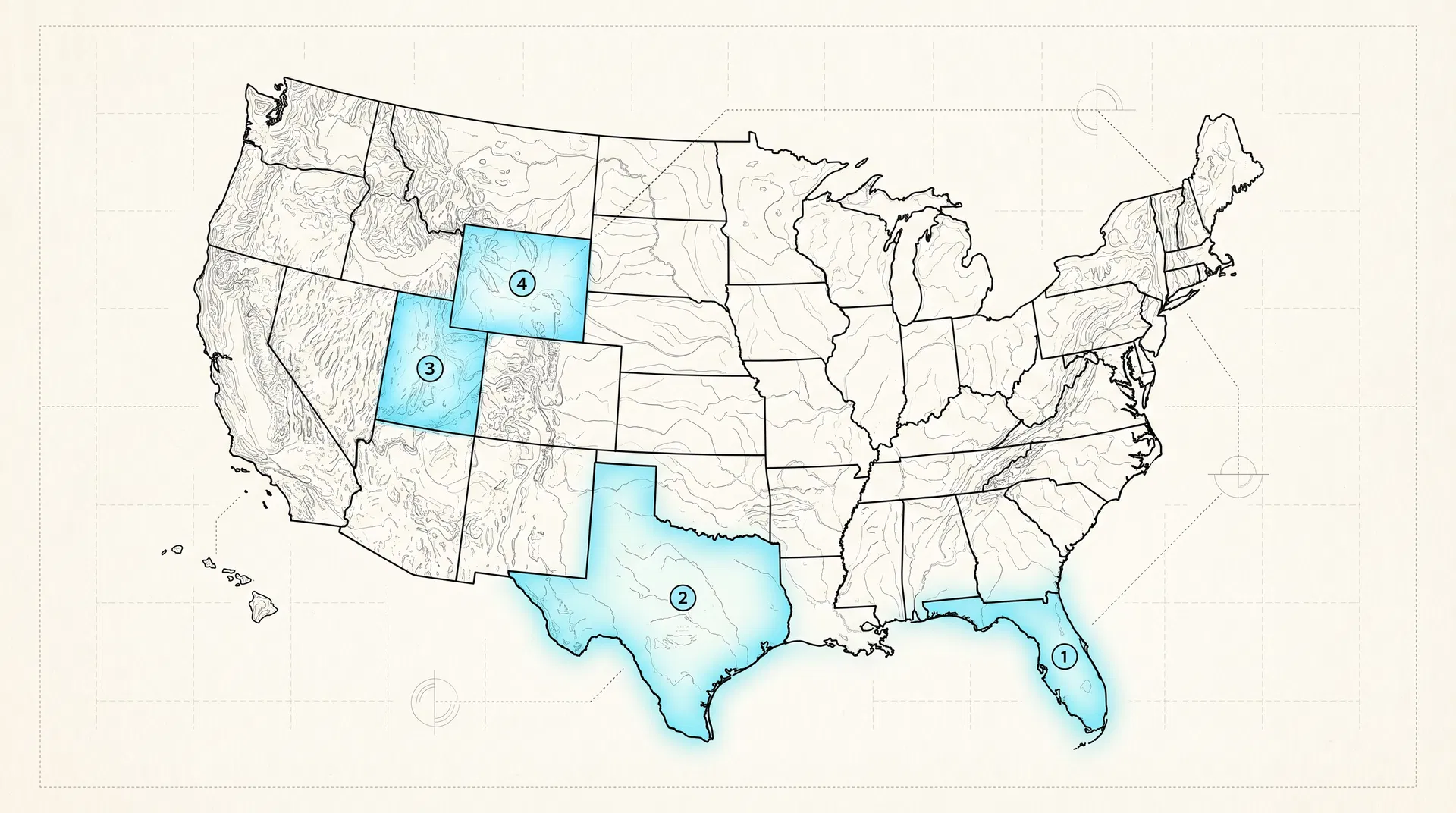
01 · FL
Florida
CS/CS/SB 1768 — Florida Statutes
02 · TX
Texas
TX HB 810 (2017), HB 3148 (2019), 22 TAC §198.6
03 · UT
Utah
Utah Code Title 58 Ch. 85 (2015), SB 199 (2024)
04 · WY
Wyoming
SF 48 — Wyoming Stem Cell Freedom Act
05 · TN
Tennessee
HB 2246 / Tenn. Code Ann. § 63-1-101 et seq.
Indications adults
ask us about.
The indications below are areas in which adults frequently consult with our network physicians. All procedures discussed are investigational and are not approved by the US Food and Drug Administration. Outcomes vary by patient and are not predictable in advance.
Knee Osteoarthritis
An investigational cellular procedure being explored for adults with knee osteoarthritis. Outcomes vary by patient and are not guaranteed.
Rotator Cuff Injury
An investigational cellular procedure being explored for adults with partial-thickness rotator cuff tendon conditions. Outcomes vary and are not guaranteed.
Chronic Back Pain
An investigational cellular procedure being explored for adults with chronic, imaging-documented low back pain. Outcomes vary and are not guaranteed.
Chronic & Diabetic Wounds
An investigational cellular wound-care procedure offered in Florida under SB 1768 in conjunction with standard wound care. Outcomes vary by patient.
Tennis & Golfer's Elbow
An investigational cellular procedure being explored for adults with chronic lateral or medial epicondylitis. Outcomes vary by patient.
Hip Osteoarthritis
An investigational cellular procedure being explored for adults with hip osteoarthritis. Outcomes vary and are not guaranteed.
From inquiry to consultation in
three deliberate steps.
01
Confidential intake
You complete a confidential health-history form. A patient advocate reviews your records, age, indication, and the relevant state framework. Submitting an intake does not establish a physician-patient relationship.
02
Physician consultation
If appropriate, we refer you to a state-licensed physician in one of the five framework states. The physician — not Select Stem Cell — independently determines whether you are a clinical candidate after their own evaluation.
03
Patient-physician decision
Any procedure, follow-up plan, and outcomes tracking is decided between you and the treating physician under their state's informed-consent and disclosure requirements. All procedures discussed are investigational.

Standards we look for
in network physicians.
Network physicians are state-licensed and operate within their state framework. Cellular products are sourced from FDA-registered establishments accredited by the AABB, AATB, NMDP, or WMDA. Each physician maintains their own informed-consent process consistent with their state's law.
- Cells sourced from FDA-registered, AABB/AATB-accredited establishments
- State-licensed physicians in their respective specialty
- Image guidance (ultrasound or fluoroscopy) where clinically indicated
- Written disclosure of investigational status before any procedure
We do not make medical claims. We do not promise outcomes. What we do is help you understand the law, the process, and your options — and connect you with a licensed physician who can evaluate your case under the framework of their state.
The Select Stem Cell standard
Truthful · Investigational · Patient-led
Patient questions,
answered honestly.
Are the stem cell procedures you describe FDA-approved?
→
No. The procedures discussed throughout this site are investigational and have not been approved by the US Food and Drug Administration for the indications described. Adult cell-based products are regulated by the FDA under 21 CFR Part 1271; certain practices fall within state statutory frameworks (Florida SB 1768, Texas HB 810/3148, Utah SB 199, Wyoming Stem Cell Freedom Act, and Tennessee HB 2246). Any procedure is a decision made between you and the treating physician.
What does Florida's SB 1768 actually do?
→
Florida's Stem Cell Therapy Act (CS/CS/SB 1768) took effect July 1, 2025. It permits Florida-licensed physicians to perform certain stem cell procedures that are not yet FDA-approved for orthopedic, wound-care, and pain-management indications, provided the cells are sourced from FDA-registered, accredited establishments and the patient receives a written disclosure of investigational status. The statute does not endorse any specific outcome.
What does Select Stem Cell actually do?
→
We are a patient-services company. We do not perform medical care, do not employ physicians, and do not own clinics. We help adults understand the law in each framework state, organize their records, and (if appropriate) refer them to a state-licensed physician for an independent consultation. The treating physician makes any clinical decision.
What is the cost of a procedure?
→
Cost varies by indication, cell product, and state. Each treating physician sets their own pricing and provides a written estimate before any procedure is scheduled. We do not collect medical fees on behalf of physicians.
Is this covered by insurance?
→
In general, no. Because the procedures discussed are investigational and not FDA-approved for these indications, most insurance plans do not cover them. Some patients use HSA/FSA funds or third-party financing. Coverage decisions are made by your insurer, not by Select Stem Cell.
Are outcomes guaranteed?
→
No. Outcomes depend on individual patient factors and are not predictable in advance. No physician in the Select Stem Cell network guarantees a specific outcome, and any testimonial or general statement on this site is not a promise of results for any other patient.
See whether a network physician
is right for your case.
The intake takes about ninety seconds. A patient advocate will review your case and respond within one business day. Submitting an intake does not establish a physician-patient relationship and does not constitute a medical recommendation.
90s
Application Time
1 day
Initial Response
$0
Application Fee
We use cookies
We use strictly necessary cookies to make this site work, and (with your permission) functional and analytics cookies to improve your experience. We do not use marketing or behavioral-advertising cookies. Read our Cookie Policy.
